The effect of methylethylpyridinol hydrochloride on the levels of α-tocopherol and transforming growth factor β-1 in patients with chronic kidney disease and type 2 diabetes mellitus
DOI:
https://doi.org/10.18413/rrpharmacology.12.763Abstract
Introduction: Chronic kidney disease (CKD) is one of the most common complications of diabetes mellitus (DM) associated with oxidative stress developing with hyperglycaemia. Excess activity of the transforming growth factor β-1 (TGF-β1) signaling pathway has been identified as a key contributor to fibrosis in the development of CKD in the context of type 2 DM.
The aim of the research was to assess the levels of α-tocopherol and TGF-β1 in individuals with CKD and type 2 DM who were undergoing standard treatment and treatment with methylethylpyridinol hydrochloride.
Materials and Methods: The study involved 60 participants with CKD and type 2 DM. The distribution of patients was carried out depending on the therapy received: the first group (n=30) used sugar-reducing agents, hypotensive, hypolipidemic and antiaggregant drugs, the second group (n=30) received methylethylpyridinol hydrochloride (10 mg once daily for 14 days) in addition to the main scheme of therapy.
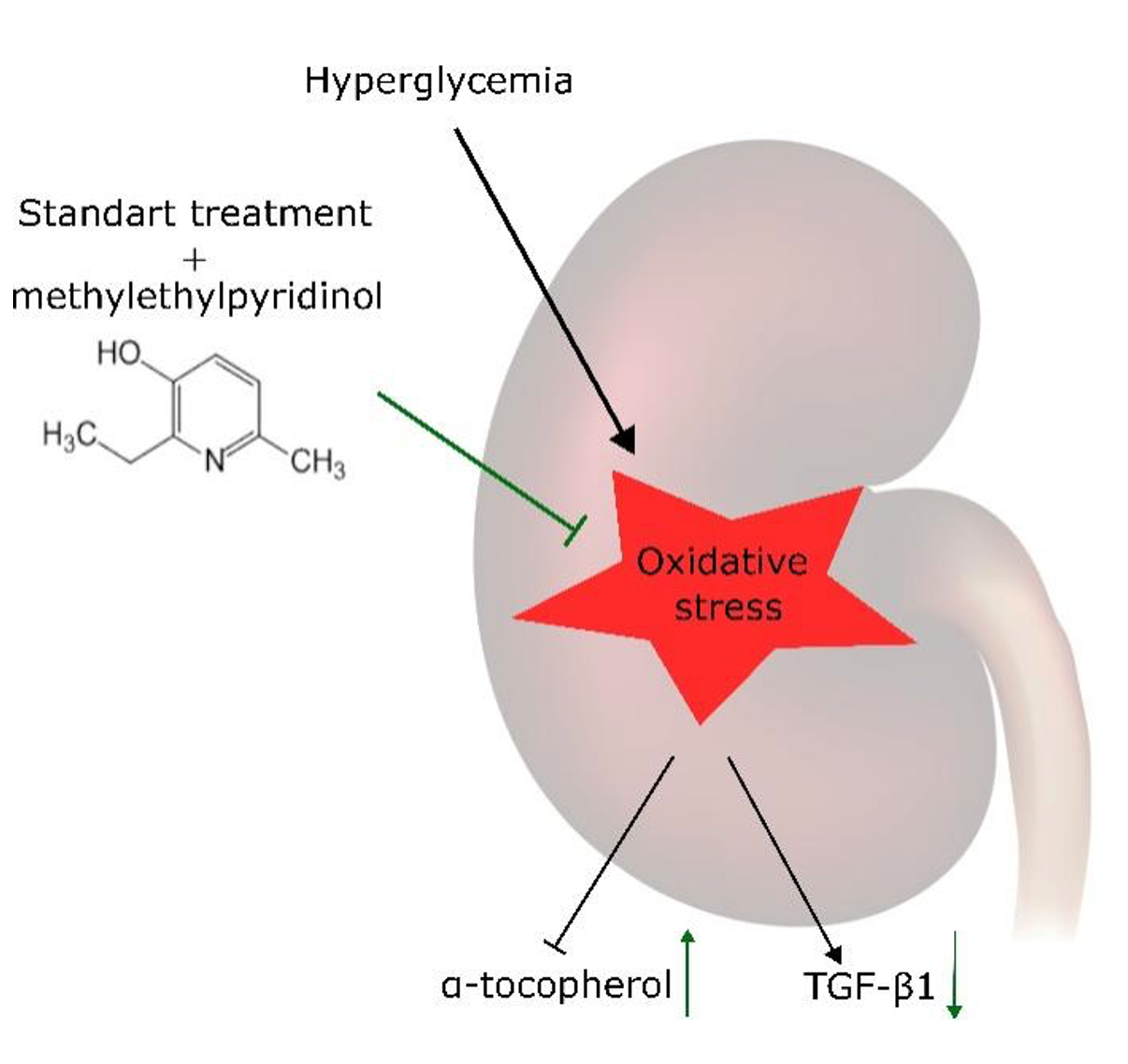
Results and Discussion: Before the initiation of inpatient treatment, all study group patients showed a 1.7-fold reduction in α-tocopherol levels. Simultaneously, the level of TGF-β1 increased by 6.1 times compared to the control group. During basic treatment, the concentration of α-tocopherol increased by 1.1 times, and the TGF-β1 level in blood serum decreased by 1.3 times compared with pre-treatment indicators. The combined treatment regimen using methylethylpyridinol hydrochloride led to a 1.4-fold increase in α-tocopherol level and a 1.6-fold reduction in TGF-β1 level.
Conclusion: Treatment with methylethylpyridinol hydrochloride has shown a significant antioxidant and anti-inflammatory effect that exceeds that of standard therapies.
Graphical Abstract
Keywords:
methylethylpyridinol hydrochloride, oxidative stress, type 2 diabetes mellitus, transforming growth factor β-1, chronic kidney disease, α-tocopherolReferences
Atukeren P (2018) Novel Prospects in Oxidative and Nitrosative Stress. InTech, UK, London, 156 pp. http://dx.doi.org/10.5772/intechopen.70102
Azzi A (2019) Tocopherols, tocotrienols and tocomonoenols: Many similar molecules but only one vitamin E. Redox Biology 26: 101259. https://doi.org/10.1016/j.redox.2019.101259[PubMed] [PMC]
Dong Y, Tong Y (2025) Lipid peroxidation in diabetic kidney disease: Mechanism and natural solution. International Journal of Molecular Sciences 26(19): 9764. https://doi.org/10.3390/ijms26199764 [PubMed] [PMC]
Galli F, Bonomini M, Bartolini D, Zatini L, Reboldi G, Marcantonini G, Gentile G, Sirolli V, Di Pietro N (2022) Vitamin E (alpha-tocopherol) metabolism and nutrition in chronic kidney disease. Antioxidants (Basel) 11(5): 989. https://doi.org/10.3390/antiox11050989 [PubMed] [PMC]
Ghafouri-Fard S, Askari A, Shoorei H, Seify M, Koohestanidehaghi Y, Hussen BM, Taheri M, Samsami M (2024) Antioxidant therapy against TGF-β/SMAD pathway involved in organ fibrosis. Journal of Cellular and Molecular Medicine 28(2): e18052. https://doi.org/1111/jcmm.18052 [PubMed] [PMC]
Gu YY, Liu XS, Huang XR, Yu XQ, Lan HY (2020) Diverse role of TGF-β in kidney disease. Frontiers in Cell and Developmental Biology 8: 123.https://doi.org/3389/fcell.2020.00123 [PubMed] [PMC]
Jin Q, Liu T, Qiao Y, Liu D, Yang L, Mao H, Ma F, Wang Y, Peng L and Zhan Y (2023) Oxidative stress and inflammation in diabetic nephropathy: Role of polyphenols. Frontiers in Immunology 14: 1185317. https://doi.org/10.3389/fimmu.2023.1185317 [PubMed] [PMC]
Kaye AD, Thomassen AS, Mashaw SA, MacDonald EM, Waguespack A, Hickey L, Singh A, Gungor D, Kallurkar A, Kaye AM, Shekoohi S, Varrassi G (2025) Vitamin E (α-Tocopherol): Emerging clinical role and adverse risks of supplementation in adults. Cureus 17(2): e78679.https://doi.org/10.7759/cureus.78679 [PubMed] [PMC]
Korneeva AV, Kurysheva NI, Loskutov IA (2025) Methylethylpyridinol in ophthalmology: Mechanisms of action and clinical results. Medical Council [Meditsinskiy Sovet] 5: 152–164. https://doi.org/10.21518/ms2025-155 [in Russian]
Liang B, Zhang XX, Li R, Zhu YC, Tian XJ, Gu N (2022) Guanxin V alleviates acute myocardial infarction by restraining oxidative stress damage, apoptosis, and fibrosis through the TGF-β1 signalling pathway. Phytomedicine 100: 154077. https://doi.org/1016/j.phymed.2022.154077 [PubMed]
Monami M, Cignarelli A, Pinto S, D'Onofrio L, Milluzzo A, Miccoli R, Penno G, Mannucci E (2021) Alpha-tocopherol and contrast-induced nephropathy: A meta-analysis of randomized controlled trials. International Journal for Vitamin and Nutrition Research 91(1-2): 188–196. https://doi.org/10.1024/0300-9831/a000573 [PubMed]
Pelle MC, Provenzano M, Busutti M, Porcu CV, Zaffina I, Stanga L, Arturi F (2022) Up-date on diabetic nephropathy. Life 12(8): 1202. https://doi.org/10.3390/life12081202 [PubMed] [PMC]
Peng D, Fu M, Wang M, Wei Y, Wei X (2022) Targeting TGF-β signal transduction for fibrosis and cancer therapy. Molecular Cancer 21(1): 104. https://doi.org/1186/s12943-022-01569-x [PubMed] [PMC]
Peresypkina A, Pazhinsky A, Pokrovskii M, Beskhmelnitsyna E, Pobeda A, Korokin M (2019) Correction of experimental retinal ischemia by l-isomer of ethylmethylhydroxypyridine malate. Antioxidants (Basel) 8(2): 34. https://doi.org/10.3390/antiox8020034 [PubMed] [PMC]
Popov SS, Anufrieva EI, Kryl’skii ED, Shulgin KK, Verevkin AN, Popova TN, Pashkov AN (2021a) The effect of methylethylpiridinol addition to the therapy on the level of pigment epithelium-derived factor and oxidative status in patients with diabetic nephropathy: randomized controlled open-label clinical study. Journal of Diabetes & Metabolic Disorders 20(1): 709–717. https://doi.org/10.1007/s40200-021-00802-6 [PubMed] [PMC]
Popov SS, Anufrieva EI, Krylskiy ED, Shulgin KK, Verevkin AN, Pashkov AN, Bolotskikh VI, Volynkina AP (2021b) The effect of combined melatonin therapy on indicators of oxidative status in chronic kidney disease developing in type 2 diabetes mellitus. Clinical Medicine 99(2): 121–127. https://doi.org/10.30629/0023-2149-2021-99-2-121-127 [in Russian]
Reiss AB, Jacob B, Zubair A, Srivastava A, Johnson M, De Leon J (2024) Fibrosis in chronic kidney disease: Pathophysiology and therapeutic targets. Journal of Clinical Medicine 13(7): 1881. https://doi.org/10.3390/jcm13071881 [PubMed] [PMC]
Swamy S, Noor SM, Mathew RO (2023) Cardiovascular disease in diabetes and chronic kidney disease. Journal of Clinical Medicine 12(22): 6984. https://doi.org/10.3390/jcm12226984
Tang PC-T, Chan AS-W, Zhang C-B, García Córdoba CA, Zhang Y-Y, To K-F, Leung K-T, Lan H-Y, Tang PM-K (2021) TGF-β1 signaling: Immune dynamics of chronic kidney diseases. Frontiers in Medicine 8: 628519. https://doi.org/3389/fmed.2021.628519 [PubMed] [PMC]
Volchegorskiĭ IA, Miroshnichenko IIu, Rassokhina LM, Faĭzullin RM, Priakhina KE (2017)Anxiolytic and antidepressant effects of emoxipine, reamberin and mexidol in experimental diabetes mellitus. S.S. Korsakov Journal of Neurology and Psychiatry [Zhurnal Nevrologii i Psikhiatrii imeni S.S. Korsakova] 117(5): 52–57. https://doi.org/10.17116/jnevro20171175152-57 [in Russian] [PubMed]
Zhao L, Zou Y, Liu F (2020) Transforming growth factor-beta1 in diabetic kidney disease. Frontiers in Cell and Developmental Biology 8: 187. https://doi.org/10.3389/fcell.2020.00187 [PubMed] [PMC]
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Anufrieva EI, Popov SS, Kryl’skii ED, Verevkin AN, Bolotskih VI, Sviridov DV

This work is licensed under a Creative Commons Attribution 4.0 International License.
 Русский
Русский
 English
English

