The C3435T genetic polymorphism of the ABCB1 (MDR1) gene as a predictor of antihypertensive efficacy of losartan monotherapy in newly diagnosed arterial hypertension
DOI:
https://doi.org/10.18413/rrpharmacology.12.1057Abstract
Introduction: Losartan, a selective angiotensin II receptor type 1 antagonist, is transported across cell membrane mediated by P-glycoprotein, the product of the ABCB1 (also known as MDR1) gene. The level of expression of this transporter protein is characterized by significant individual variability, caused by genetic factors. Well-known polymorphisms C3435T and C1236T in the ABCB1 gene potentially could affect a functional activity of the P-glycoprotein, thus modulating the pharmacokinetic parameters and clinical efficacy of the substrates of this transporter, including losartan. Therefore, the aim of this research is to indicate the correlation between common SNPs C3435T and C1236T of the MDR1 gene and the efficacy of 6-week losartan monotherapy course in patients with a newly diagnosed arterial hypertension (AH).
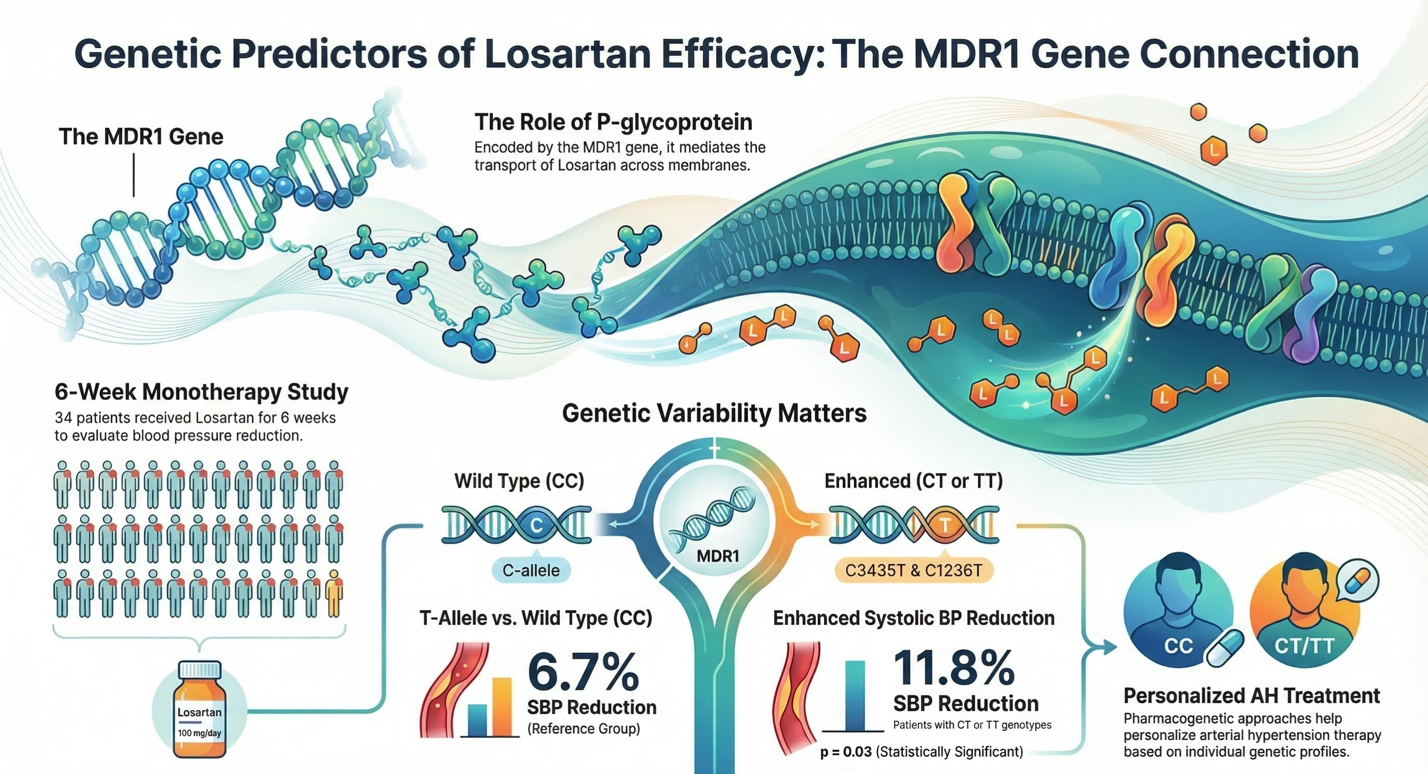
Materials and Methods: The study included 34 patients (70.6% women, mean age 48.3 ± 7.4 years). All participants were given losartan 100 mg/day for 6 weeks. Genotyping for C3435T and C1236T polymorphisms was made by using allele-specific PCR with electrophoretic detection. The efficacy of the therapy was evaluated by the reduction of systolic (SBP) and diastolic (DBP) blood pressure from the reference level.
Results: Patients with CT or TT genotypes for the C3435T polymorphism showed a statistically meaningful, greater reduction in SBP (11.8% ± 9.7) compared to homozygous CC genotype (”wild” type; 6.7% ± 9.6; p=0.03). No significant differences were found in SBP reduction for the C1236T polymorphism (p=0.07). Changes in DBP did not correlate with either of the studied polymorphisms.
Conclusions: The C3435T genetic polymorphism of the MDR1 gene is a potential predictor of the efficacy of losartan antihypertensive therapy. The carriers of the T allele (CT/TT genotypes) demonstrate more expressed hypotensive response, which may be caused by modulation of tissue distribution of the drug or its interaction with endogenous systems (ouabain) regulating blood pressure. The obtained data highlights the importance of a pharmacogenetic approach for personalizing AH treatment.
Graphical Abstract
Keywords:
P-glycoprotein, ABCB1, MDR1, genetic polymorphism, arterial hypertension, losartan, personalized medicineReferences
Goktas MT, Pepedil F, Karaca O, Kalkisim S, Cevik L, Gumus E, Guven GS, Babaoglu MO, Bozkurt A, Yasar U (2016) Relationship between genetic polymorphisms of drug efflux transporter MDR1 (ABCB1) and response to losartan in hypertension patients. European Review Medical and Pharmacological Sciences 20(12): 2460–2467. [PubMed]
Sinitsina II, Boyarko AV, Temirbulatov II, Mirzaev KB, Grishina EA, Sozaeva ZA, Akmalova KA, Shuev GN, Denisenko NP, Kachanova AA, Sychev DA (2021) The influence of CYP2C9 gene polymorphisms on the efficacy of losartan in patients with grade I–II arterial hypertension. Pharmateka 28(3): 57–61. https://doi.org/10.18565/pharmateca.2021.3.57-61
Soldner A, Benet LZ, Mutschler E, Christians U (2000) Active transport of the angiotensin-II antagonist losartan and its main metabolite EXP 3174 across MDCK-MDR1 and caco-2 cell monolayers. British Journal of Pharmacology 129: 1235–1243. https://doi.org/10.1038/sj.bjp.0703150 [PubMed] [PMC]
Sychev DA (2021) (Ed) Applied Pharmacogenetics. Moscow-Tver: Triada Publishing House, 496 pp.
Tripodi G, Citterio L, Kouznetsova T, Lanzani C, Florio M, Modica R, Messaggio E, Hamlyn JM, Zagato L, Bianchi G, Staessen JA, Manunta P (2009) Steroid biosynthesis and renal excretion in human essential hypertension: association with blood pressure and endogenous ouabain. American Journal of Hypertension 22(4): 357–363. https://doi.org/10.1038/ajh.2009.3[PubMed] [PMC]
Yasar U, Babaoglu MO, Bozkurt A (2008) Disposition of a CYP2C9 phenotyping agent, losartan, is not influenced by the common 3435C>T variation of the drug transporter gene ABCB1 (MDR1). Basic & Clinical Pharmacology & Toxicology 103: 176–179. https://doi.org10.1111/j.1742-7843.2008.00283.x [PubMed]
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Shelekhova TV, Ruta АV, Luchinina ЕV, Bontsevich RA, Zaitсeva MR, Lewanov AN, Сherednikova PА, Luchinin EА

This work is licensed under a Creative Commons Attribution 4.0 International License.
 Русский
Русский
 English
English

