Evaluation of pharmacological correction of hypoxic-ischemic encephalopathy sequelae using peptide erythropoietin analogs in a mouse model of mild and moderate hypoxic-ischemic encephalopathy during the late remodeling phase
DOI:
https://doi.org/10.18413/rrpharmacology.12.1075Abstract
Introduction: Hypoxic-ischemic encephalopathy (HIE) is an early-life brain injury that remains a leading cause of long-term neurodevelopmental deficits, including cerebral palsy, epilepsy, and cognitive-behavioral impairment. Because persistent neuroinflammation and progressive neurodegeneration contribute to delayed outcomes, targeting EPOR-CD131 with nonerythropoietic erythropoietin derivatives represents a promising strategy to modulate neurovascular unit function and limit secondary injury beyond the acute therapeutic window.
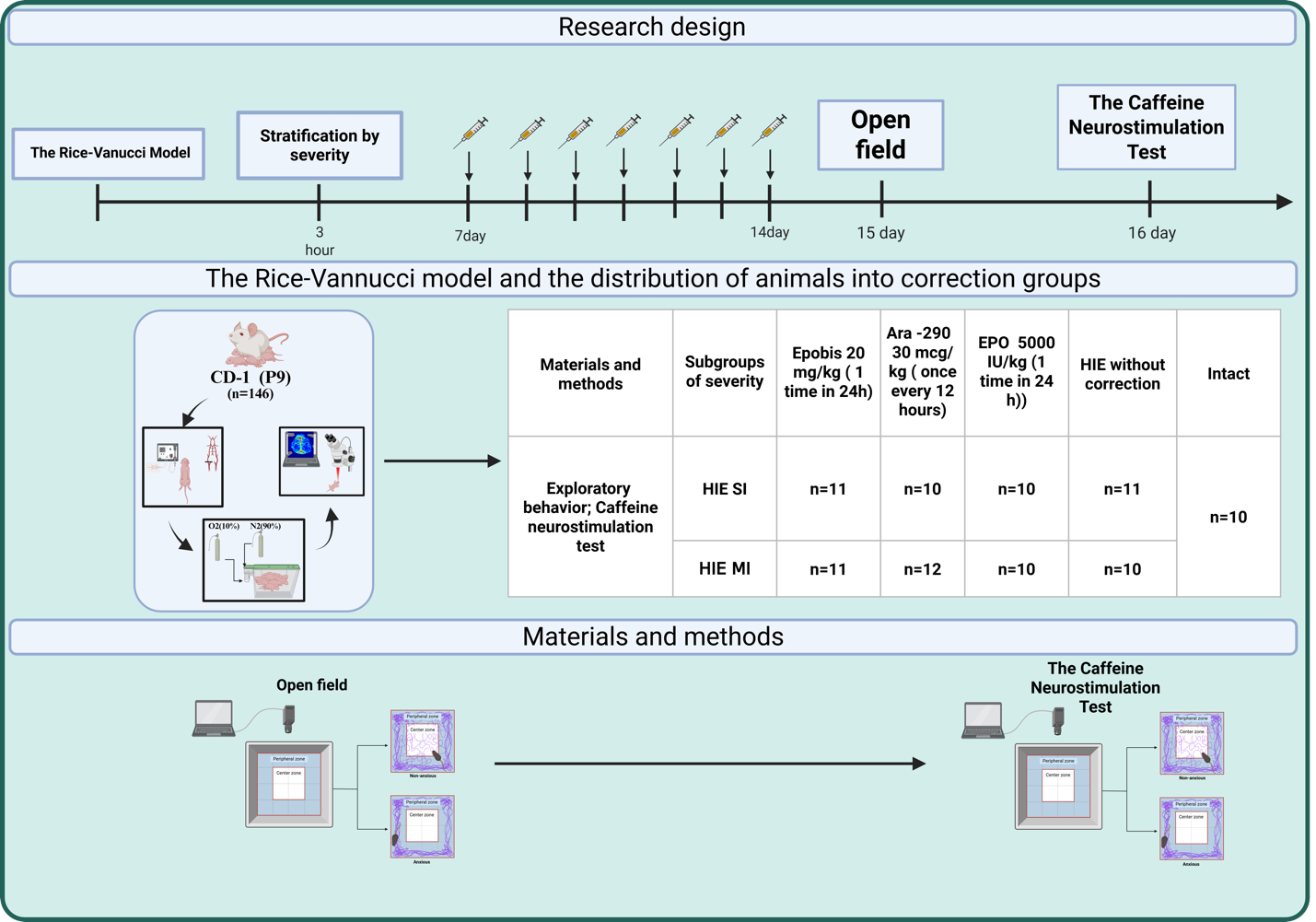
Materials and Methods: CD-1 mice underwent neonatal hypoxia–ischemia at postnatal day 9 (modified Rice–Vannucci) and were stratified 3 h later into mild or moderate injury using laser speckle imaging (RFLSI-ZW), then allocated to treatment groups. Starting on day 7 post-injury, EPO (5000 IU/kg) and Epobis (20 mg/kg) were administered subcutaneously once daily and Ara-290 (30 µg/kg) intraperitoneally twice daily for 7 days, after which exploratory behavior and caffeine-induced neurostimulation responses were evaluated.
Results: In this experimental study, Epobis and Ara-290 normalized mild HIE hyperlocomotion in the open field, whereas EPO mainly improved anxiety-like behavior and only Epobis increased peripheral time toward intact levels. In the caffeine challenge, all treatments reversed paradoxical suppression in mild HIE, and Epobis most effectively reduced caffeine-induced hyperreactivity and restored baseline activity in moderate HIE.
Conclusion: Epobis and Ara-290 normalized mild HIE hyperlocomotion in the open field, whereas Epobis provided the most comprehensive behavioral correction and EPO mainly improved anxiety-related measures. All treatments restored a physiological caffeine response in mild HIE, and Epobis most effectively improved behavior and reduced caffeine-induced hyperreactivity in moderate HIE, supporting further preclinical and subsequent clinical evaluation.
Graphical Abstract
Keywords:
hypoxic-ischemic encephalopathy, exploratory behavior, caffeine neurostimulation test, Epobis, Ara-290, EPO, laser speckle contrast imagingReferences
Amoruso P, Lecce E, Scotto di Palumbo A, Sacchetti M, Bazzucchi I (2026) Caffeine as an ergogenic aid for neuromuscular performance: Mechanisms of action from brain to motor units. Nutrients 18(2): 252. https://doi.org/10.3390/nu18020252 [PubMed] [PMC]
Anusornvongchai T, Nangaku M, Jao TM, Wu CH, Ishimoto Y, Maekawa H, Tanaka T, Shimizu A, Yamamoto M, Suzuki N, Sassa R, Inagi R (2018) Palmitate deranges erythropoietin production via transcription factor ATF4 activation of unfolded protein response. Kidney International 94(3): 536–550. https://doi.org/10.1016/j.kint.2018.03.011 [PubMed]
Arnautovic T, Sinha S, Laptook AR (2024) Neonatal hypoxic-ischemic encephalopathy and hypothermia treatment. Obstetrics & Gynecology 143(1): 67–81. https://doi.org/10.1097/AOG.0000000000005392 [PubMed] [PMC]
Boison D, Sandau US, Ruskin DN, Kawamura M Jr, Masino SA (2013) Homeostatic control of brain function – new approaches to understand epileptogenesis. Frontiers in Cellular Neuroscience 7: 109. https://doi.org/10.3389/fncel.2013.00109 [PubMed] [PMC]
Carbone MG, Pagni G, Tagliarini C, Maremmani I, Maremmani AGI (2025) Caffeine in aging brains: Cognitive enhancement, neurodegeneration, and emerging concerns about addiction. International Journal of Environmental Research and Public Health 22(8): 1171. https://doi.org/10.3390/ijerph22081171 [PubMed] [PMC]
Chakkarapani E, de Vries LS, Ferriero DM, Gunn AJ (2025) Neonatal encephalopathy and hypoxic-ischemic encephalopathy: The state of the art. Pediatric Research 98(7): 2444–2458. https://doi.org/10.1038/s41390-025-03986-2 [PubMed] [PMC]
Chan NH, Hawkins CC, Rodrigues, BV, Cornet MC, Gonzalez FF, Wu YW (2025) Neuroprotection for neonatal hypoxic-ischemic encephalopathy: A review of novel therapies evaluated in clinical studies. Developmental Medicine & Child Neurology 67(5): 591–599. https://doi.org/10.1111/dmcn.16184 [PubMed]
Cizmeci MN, Christensen R, van Steenis A, de Vries LS (2025) Neuroprognostication in neonatal encephalopathy due to presumed hypoxic-ischemic encephalopathy. Pediatric Research 98(7): 2459–2470. https://doi.org/10.1038/s41390-025-04058-1 [PubMed]
Dmytriyeva O, Pankratova S, Korshunova I, Walmod PS (2016) Epobis is a nonerythropoietic and neuroprotective agonist of the erythropoietin receptor with anti-inflammatory and memory enhancing effects. Mediators of Inflammation 2016: 1346390. https://doi.org/10.1155/2016/1346390 [PubMed] [PMC]
Götz J, Wieters F, Fritz VJ, Käsgen O, Kalantari A, Fink GR, Aswendt M (2023) Temporal and spatial gene expression profile of stroke recovery genes in mice. Genes 14(2): 454. https://doi.org/10.3390/genes14020454 [PubMed] [PMC]
Gunnarson E, Song Y, Kowalewski JM, Brismar H, Brines M, Cerami A, Andersson U, Zelenina M, Aperia A (2009) Erythropoietin modulation of astrocyte water permeability as a component of neuroprotection. Proceedings of the National Academy of Sciences of the United States of America 106(5): 1602–1607. https://doi.org/10.1073/pnas.0812708106 [PubMed] [PMC]
Håkansson G, Robertsson Grossmann K, Ådén U, Blennow M, Fransson P (2025) Functional brain connectivity in early adolescence after hypothermia-treated neonatal hypoxic-ischemic encephalopathy. Pediatric Research 98(5): 1827–1834. https://doi.org/10.1038/s41390-025-03951-z [PubMed] [PMC]
Huntingford SL, Boyd SM, McIntyre SJ, Goldsmith SC, Hunt RW, Badawi N (2024) Long-term outcomes following hypoxic ischemic encephalopathy. Clinics in Perinatology 51(3): 683–709. https://doi.org/10.1016/j.clp.2024.04.008 [PubMed]
Juul SE, Yachnis AT, Rojiani AM, Christensen RD (1999) Immunohistochemical localization of erythropoietin and its receptor in the developing human brain. Pediatric and Developmental Pathology 2(2): 148–158. https://doi.org/10.1007/s100249900103 [PubMed]
Konrad J, Chau V, Selvanathan T (2025) Advances in neonatal hypoxic-ischemic encephalopathy. Pediatric Clinics of North America 72(5): 939–955. https://doi.org/10.1016/j.pcl.2025.05.006 [PubMed]
Lakatos A, Kolossváry M, Szabó M, Jermendy Á, Barta H, Gyebnár G, Rudas G, Kozák LR (2019) Neurodevelopmental effect of intracranial hemorrhage observed in hypoxic ischemic brain injury in hypothermia-treated asphyxiated neonates – an MRI study. BMC Pediatrics 19(1): 430. https://doi.org/10.1186/s12887-019-1777-z [PubMed] [PMC]
Lan KM, Tien LT, Cai Z, Lin S, Pang Y, Tanaka S, Rhodes PG, Bhatt AJ, Savich RD, Fan LW (2016) Erythropoietin ameliorates neonatal hypoxia-ischemia-induced neurobehavioral deficits, neuroinflammation, and hippocampal injury in the juvenile rat. International Journal of Molecular Sciences 17(3): 289. https://doi.org/10.3390/ijms17030289 [PubMed] [PMC]
Mike JK, White Y, Ha J, Iranmahboub A, Hawkins C, Hutchings RS, Vento C, Manzoor H, Wang A, Goudy BD, Vali P, Lakshminrusimha S, Gobburu JVS, Long-Boyle J, Fineman JR, Ferriero DM, Maltepe E (2024) Perinatal caffeine administration improves outcomes in an ovine model of neonatal hypoxia-ischemia. Stroke 55(11): 2705–2715. https://doi.org/10.1161/STROKEAHA.124.048264 [PubMed] [PMC]
Mills JH, Kim DG, Krenz A, Chen JF, Bynoe MS (2012) A2A adenosine receptor signaling in lymphocytes and the central nervous system regulates inflammation during experimental autoimmune encephalomyelitis. Journal of Immunology 188(11): 5713–5722. https://doi.org/10.4049/jimmunol.1200545 [PubMed] [PMC]
Montaldo P, Cirillo M, Burgod C, Caredda E, Ascione S, Carpentieri M, Puzone S, D'Amico A, Garegrat R, Lanza M, Moreno Morales M, Atreja G, Shivamurthappa V, Kariholu U, Aladangady N, Fleming P, Mathews A, Palanisami B, Windrow J, Harvey K, Soe A, Pattnayak S, Sashikumar P, Harigopal S, Pressler R, Wilson M, De Vita E, Shankaran S, Thayyil S; COMET Trial Group (2024) Whole-body hypothermia vs targeted normothermia for neonates with mild encephalopathy: A multicenter pilot randomized clinical trial. JAMA Network Open 7(5): e249119. https://doi.org/10.1001/jamanetworkopen.2024.9119 [PubMed] [PMC]
Ostrowski D, Heinrich R (2018) Alternative erythropoietin receptors in the nervous system. Journal of Clinical Medicine 7(2): 24. https://doi.org/10.3390/jcm7020024 [PubMed] [PMC]
Pankratova S, Gu B, Kiryushko D, Korshunova I, Køhler LB, Rathje M, Bock E, Berezin V (2012) A new agonist of the erythropoietin receptor, Epobis, induces neurite outgrowth and promotes neuronal survival. Journal of Neurochemistry 121(6): 915–923. https://doi.org/10.1111/j.1471-4159.2012.07751.x [PubMed]
Pokrovskii V, Lapin K, Antonova V, Korokin M, Gudyrev O, Gureev V, Korokina L, Scheblykina O, Nesterov A, Maslinikova M, Chatsky I, Mukhamedov D, Pokrovskii M (2026) Use of laser speckle contrast imaging for distribution of animals by severity of brain tissue damage in a neonatal hypoxia-ischemia model in mice. Brain Sciences 16: 102. https://doi.org/10.3390/brainsci16010102 [PubMed] [PMC]
Pokrovsky VM, Chatsky IV, Maslinikova MR, Zabolotskikh NV, Nesterov AV, Popov NV, Korokin MV (2025) Investigation of behavioral characteristic changes in animals with varying injury severity following brain tissue damage in a mouse model of neonatal hypoxia-ischemia during the juvenile period. Research Results in Pharmacology 11(4): 282–292. https://doi.org/10.18413/rrpharmacology.11.863
Ramirez A, Peyvandi S, Cox S, Gano D, Xu D, Tymofiyeva O, McQuillen PS (2022) Neonatal brain injury influences structural connectivity and childhood functional outcomes. PLOS ONE 17(1): e0262310. https://doi.org/10.1371/journal.pone.0262310 [PubMed] [PMC]
Sheldon RA, Windsor C, Ferriero DM (2018) Strain-related differences in mouse neonatal hypoxia-ischemia. Developmental Neuroscience 40(5–6): 490–496. https://doi.org/10.1159/000495880 [PubMed] [PMC]
Simon F, Floros N, Ibing W, Schelzig H, Knapsis (2019) Neurotherapeutic potential of erythropoietin after ischemic injury of the central nervous system. Neural Regeneration Research 14(8): 1309–1312. https://doi.org/10.4103/1673-5374.253507 [PubMed] [PMC]
Spencer APC, Lee-Kelland R, Brooks JCW, Jary S, Tonks J, Cowan FM, Thoresen M, Chakkarapani E (2023) Brain volumes and functional outcomes in children without cerebral palsy after therapeutic hypothermia for neonatal hypoxic-ischaemic encephalopathy. Developmental Medicine & Child Neurology 65(3): 367–375. https://doi.org/10.1111/dmcn.15369 [PubMed] [PMC]
Sturgess JE, Ting-A-Kee RA, Podbielski D, Sellings LH, Chen JF, van der Kooy (2010) Adenosine A1 and A2A receptors are not upstream of caffeine’s dopamine D2 receptor-dependent aversive effects and dopamine-independent rewarding effects. European Journal of Neuroscience 32(1): 143–154. https://doi.org/10.1111/j.1460-9568.2010.07247.x [PubMed] [PMC]
Sun H, Gonzalez F, McQuillen PS (2020) Caffeine restores background EEG activity independent of infarct reduction after neonatal hypoxic ischemic brain injury. Developmental Neuroscience 42(1): 72–82. https://doi.org/10.1159/000509365 [PubMed] [PMC]
Suresh S, Rajvanshi PK, Noguchi CT (2020) The many facets of erythropoietin physiologic and metabolic response. Frontiers in Physiology 10: 1534. https://doi.org/10.3389/fphys.2019.01534 [PubMed] [PMC]
Ureña-Guerrero ME, Castañeda-Cabral JL, Rivera-Cervantes MC, Macias-Velez RJ, Jarero-Basulto JJ, Gudiño-Cabrera G, Beas-Zárate (2020) Neuroprotective and neurorestorative effects of Epo and VEGF: Perspectives for new therapeutic approaches to neurological diseases. Current Pharmaceutical Design 26(12): 1263–1276. https://doi.org/10.2174/1381612826666200114104342 [PubMed]
Wang R, Wu X, Zhao H, Min L, Tao Z, Ji X, Luo Y (2016) Effects of erythropoietin combined with tissue plasminogen activator on the rats following cerebral ischemia and reperfusion. Brain Circulation 2(1): 54–60. https://doi.org/10.4103/2394-8108.178552 [PubMed] [PMC]
Wang RL, Yang ZH, Huang YY, Hu Y, Wang YL, Yan F, Zheng YM, Han ZP, Fan JF, Tao Z, Zhao HP, Li SJ, Luo YM (2024) Erythropoietin-derived peptide ARA290 mediates brain tissue protection through the beta-common receptor in mice with cerebral ischemic stroke. CNS Neuroscience & Therapeutics 30(3): e14676. https://doi.org/10.1111/cns.14676 [PubMed] [PMC]
Wu YW, Monsell SE, Glass HC, Wisnowski JL, Mathur AM, McKinstry RC, Bluml S, Gonzalez FF, Comstock BA, Heagerty PJ, Juul SE (2023) How well does neonatal neuroimaging correlate with neurodevelopmental outcomes in infants with hypoxic-ischemic encephalopathy? Pediatric Research 94(3): 1018–1025. https://doi.org/10.1038/s41390-023-02510-8 [PubMed] [PMC]
Xiong T, Yang X, Qu Y, Chen H, Yue Y, Wang H, Zhao F, Li S, Zou R, Zhang L, Mu D (2019) Erythropoietin induces synaptogenesis and neurite repair after hypoxia ischemia-mediated brain injury in neonatal rats. NeuroReport 30(11): 783–789. https://doi.org/10.1097/WNR.0000000000001285 [PubMed]
Yang M, Wang K, Liu B, Shen Y, Liu G (2025) Hypoxic-ischemic encephalopathy: Pathogenesis and promising therapies. Molecular Neurobiology 62(2): 2105–2122. https://doi.org/10.1007/s12035-024-04398-9 [PubMed]
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Pokrovsky VM

This work is licensed under a Creative Commons Attribution 4.0 International License.
 Русский
Русский
 English
English

