Platform for preclinical screening of neuroprotective strategies: a clinically relevant ICH model and standardized functional test battery
DOI:
https://doi.org/10.18413/rrpharmacology.12.1043Abstract
Introduction: Intracerebral hemorrhage (ICH) is among the most severe forms of stroke. Whereas traditional hemorrhagic stroke models reproduce hemorrhage within the striatum (specifically the putamen), localization of the hematoma with involvement of the internal capsule and globus pallidus may better recapitulate clinically relevant sensorimotor deficits due to more extensive basal ganglia involvement and damage to dense clusters of white matter tracts.
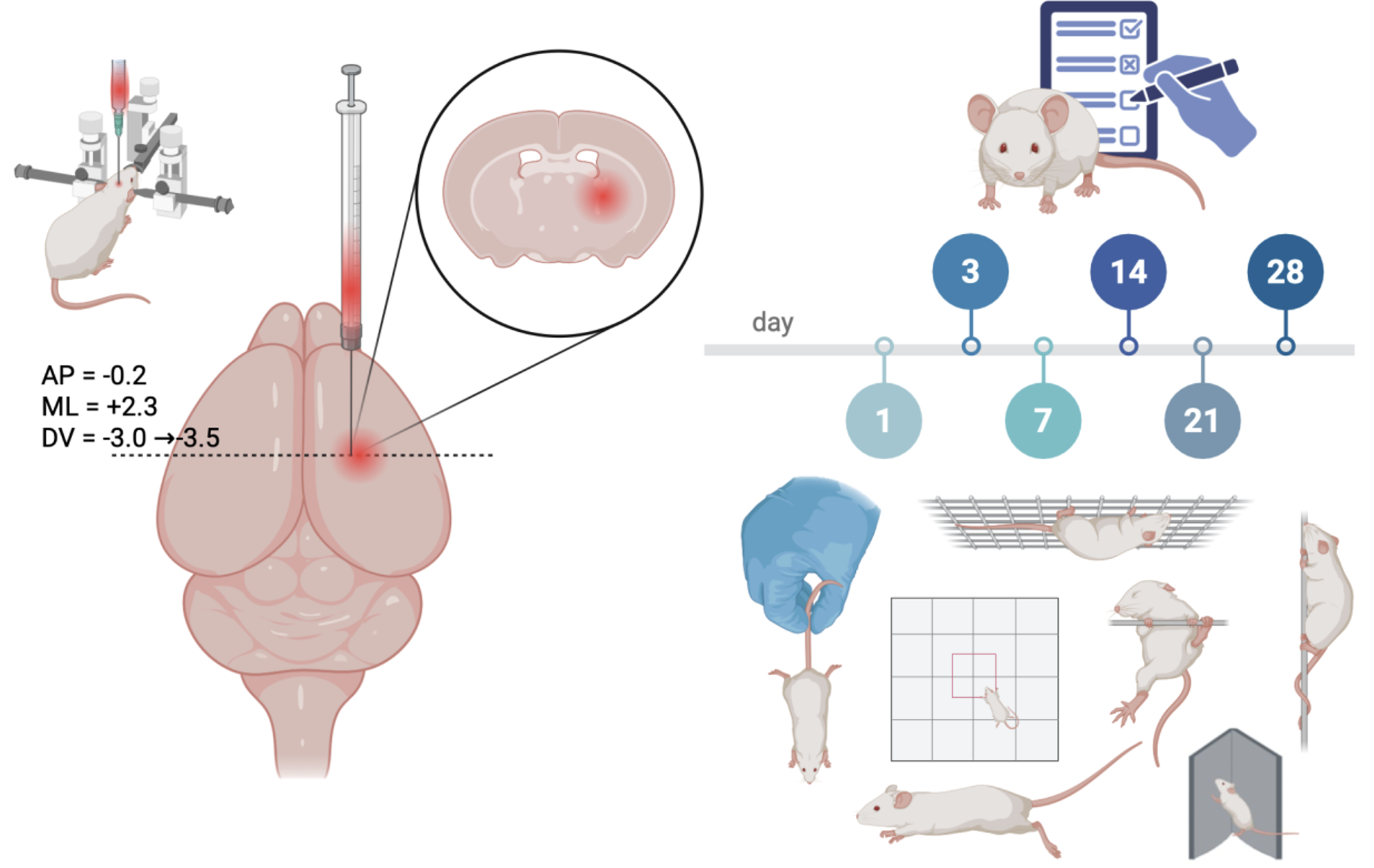
Materials and Methods: Male CD1 mice (3–4 months, 30–35 g) were assigned to intact control (n=15) or ICH (n=15) groups. ICH was induced by stereotaxic injection of 30 μL autologous blood into the right cerebral hemisphere (2.3 mm lateral, 0.2 mm caudal to bregma, depths 3.0 and 3.5 mm). Neurological status was assessed using the modified Garcia scale, limb placement tests, horizontal and vertical pole tests, inverted grid test, corner test, and open field test at baseline and on days 1, 3, 7, 14, 21, and 28.
Results and Discussion: The model showed high reproducibility with 33–40% acute mortality. All ICH mice developed contralateral sensorimotor-pyramidal deficits with predominant forelimb involvement. Garcia scores declined from 21 to 14.83±1.72 on day 1 with gradual but incomplete recovery by day 28 (18 [18;18.5]). Forelimb placement showed >50% acute deficit with a nonlinear recovery pattern, whereas hindlimb deficits appeared later (days 3–7) and persisted to day 21. The modified Garcia scale, forelimb placement, modified horizontal bar, and open field tests were most sensitive to functional impairment and recovery.
Conclusion: Stereotaxic autologous blood injection into the basal ganglia (putamen and globus pallidus) with involvement of the internal capsule in mice reproduces clinically relevant, reproducible sensorimotor deficits with persistent functional impairment, making this model suitable for preclinical testing of neuroprotective strategies.
Graphical Abstract
Keywords:
hemorrhagic stroke, internal capsule, ICH model, mice, Garcia scale, behavioral assessment, sensorimotor deficitReferences
Bachour SP, Hevesi M, Bachour O, Sweis BM, Mahmoudi J, Brekke JA, Divani AA (2016) Comparisons between Garcia, Modo, and Longa rodent stroke scales: Optimizing resource allocation in rat models of focal middle cerebral artery occlusion. Journal of the Neurological Sciences 364: 136-140. https://doi.org/10.1016/j.jns.2016.03.029 [PubMed]
Balkaya M, Kröber JM, Rex A, Endres M (2013) Assessing post-stroke behavior in mouse models of focal ischemia. Journal of Cerebral Blood Flow and Metabolism 33 (3): 330–338. https://doi.org/10.1038/jcbfm.2012.185 [PubMed] [PMC]
Chung CS, Caplan LR, Yamamoto Y, Chang HM, Lee SJ, Song HJ, Lee HS, Shin HK, Yoo KM (2000) Striatocapsular aemorrhage. Brain 123 (Pt 9): 1850–1862. https://doi.org/10.1093/brain/123.9.1850 [PubMed]
Gupta S, Xiao M, Liu N, Zhao Y, Zhao X, Huang Y, Wu Y, Lin Z, Ji Z, Xu H, Zhu M, Pan S, Huang K (2025) Involvement of the posterior limb of the internal capsule independently predicts the prognosis of patients with basal ganglia and thalamic hemorrhage. Frontiers in Neurology 15: 1475444. https://doi.org/10.3389/fneur.2024.1475444 [PubMed] [PMC]
Krafft PR, Rolland WB, Duris K, Lekic T, Campbell A, Tang J, Zhang JH (2012) Modeling intracerebral hemorrhage in mice: injection of autologous blood or bacterial collagenase. Journal of Visualized Experiments 67: e4289. https://doi.org/10.3791/4289 [PubMed] [PMC]
Liu Y, Lu G, Su XW, Ding T, Wang WL, Li YM, Poon WS, Ao LJ (2018) Characterization of axon damage, neurological deficits, and histopathology in two experimental models of intracerebral hemorrhage. Frontiers in Neuroscience 12: 928. https://doi.org/10.3389/fnins.2018.00928 [PubMed] [PMC]
Matsumura K, Kumar TP, Guddanti T, Yan Y, Blackburn SL, McBride DW (2019) Neurobehavioral deficits after subarachnoid hemorrhage in mice: sensitivity analysis and development of a new composite score. Journal of the American Heart Association 8 (8): e011699.https://doi.org/10.1161/JAHA.118.011699 [PubMed] [PMC]
Matsushita H, Hijioka M, Hisatsune A, Isohama Y, Iwamoto S, Terasawa H, Katsuki H (2013) MRI-based analysis of intracerebral hemorrhage in mice reveals relationship between hematoma expansion and the severity of symptoms. PLoS ONE 8 (7): e67691. https://doi.org/10.1371/journal.pone.0067691 [PubMed] [PMC]
Nesterova NI, Shcheblykina OV, Kolesnichenko PD, Nesterov AV, Shcheblykin DV, Popova IA, Yakovlev DV (2019) Neuroprotective effects of taurine and 3-hydroxypyridine derivatives in the intracerebral hemorrhage model in rats. Research Results in Pharmacology 5(3): 87–94. https://doi.org/10.3897/rrpharmacology.5.36988
Paxinos G, Franklin KBJ (2012) The Mouse Brain in Stereotaxic Coordinates, 4th Academic Press, San Diego, 360 pp.
Ruan J, Yao Y (2020) Behavioral tests in rodent models of stroke. Brain Hemorrhages 1 (4): 171–184. https://doi.org/10.1016/j.hest.2020.09.001 [PubMed] [PMC]
Shcheblykina OV, Shcheblykin DV, Trunov KS, Danilenko AP, Lipatov VS (2022) Experimental study of new derivatives of 3-hydroxypyridine as pharmacological agents for the correction of ischemic brain injury after intracerebral hemorrhage. Research Results in Pharmacology 8 (1): 71–83. https://doi.org/10.3897/rrpharmacology.8.8037
Young CB, Reddy V, Sonne J (2025) Neuroanatomy, Basal Ganglia. In: StatPearls [Internet]. StatPearls Publishing, Treasure Island (FL). Available from: https://www.ncbi.nlm.nih.gov/books/NBK537141/
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Deev RV, Shcheblykina OV, Chao Z, Kostina DA, Sun W, Gureev VV, Slepov IuK, Zhunusov NS, Korokina LV, Gudyrev OS, Nekrasova AA, Osipova OA, Repina EI, Nesterov AV, Pokrovskaya TG

This work is licensed under a Creative Commons Attribution 4.0 International License.
 Русский
Русский
 English
English

