Влияние производного азоло-триазинов соединения АВ-19 на развитие диабетической кардиомиопатии у крыс
DOI:
https://doi.org/10.18413/rrpharmacology.12.1049Аннотация
Введение: В структуре смертности при СД доминируют заболевания сердечно-сосудистой этиологии, включая кардиомиопатии, связанные с дисфункцией эндотелия и нарушением гликирования белков. В статье представлены данные по выявлению функциональных и структурных изменений, происходящих при формировании диабетической кардиомиопатии и изучение кардиопротекторных эффектов антигликирующих веществ – аминогуанидина и соединения АВ-19.
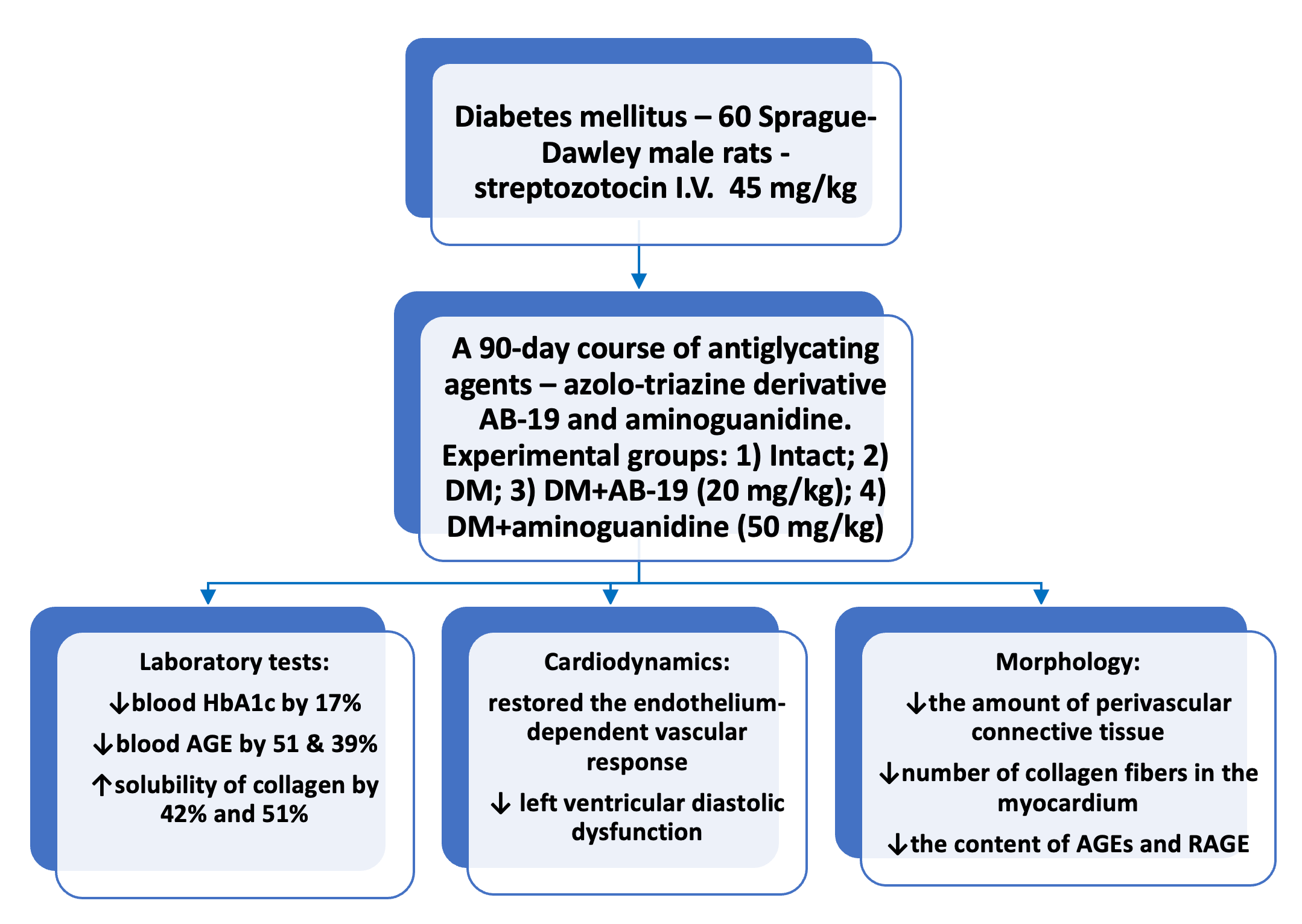
Материалы и методы: Диабетическую кардиомиопатию моделировали стрептозотоцином в дозе 45 мг/кг. Наблюдения проводили в течение 12 недель. Фиксировали уровень глюкозы и концентрацию гликозилированного гемоглобина в крови, по истечению срока формирования диабетической кардиомиопатии были изучены эндотелиопротекторные свойства; сократительная активность сердца; растворимость коллагена сухожилий хвоста, выполнены морфологические исследования сердца и сосудов миокарда.
Результаты: Соединение АВ-19 (20 мг/кг) и аминогуанидин (50 мг/кг) при пероральном курсовом введении животным с экспериментальным СД снижали HbA1c в крови на 17% по сравнению с контрольными диабетическими крысами, ограничивали прирост уровня КПГ в крови на 51 и 39%, improved the solubility of collagen by 42% and 51%, respectively, восстанавливали эндотелий-зависимую реакцию сосудов, уменьшали проявления диастолической дисфункции левого желудочка, связанной с гипертрофией и фиброзом миокарда у животных с СД, что подтверждалось морфологически - у животных, получавших соединение АВ-19 и аминогуанидин, снижалось количество периваскулярной соединительной ткани, а также сокращалось количество коллагеновых волокон в миокарде, уменьшалось содержание иммунореактивного материала при ИГХ исследовании с использованием первичных антител против AGEs и RAGE относительно контрольной группы сахарного диабета.
Заключение: Соединение АВ-19 снижает функциональные и структурные проявления диабетической кардиомиопатии.
Графическая аннотация
Ключевые слова:
азоло-триазины, антигликация, поздние осложнения сахарного диабета, диабетическая кардиомиопатияБиблиографические ссылки
Avtandilov GG (2002) Fundamentals of Quantitative Pathological Anatomy. Medicine Publishers, Moscow, 240 pp. [in Russian]
Bagriy AE, Suprun EV, Mikhaylichenko ES, Golodnikov IA (2020) Heart failure and type 2 diabetes: current state of the problem. Russian Journal of Cardiology [Rossijskij Kardiologicheskij Zhurnal] 25(4): 79–85. https://doi.org/10.15829/1560-4071-2020-3858 [in Russian]
Bolton WK, Cattran DC, Williams ME, Adler SG, Appel GB, Cartwright K, Foiles PG, Freedman BI, Raskin P, Ratner RE, Spinowitz BS, Whittier FC, Wuerth JP; ACTION I Investigator Group (2004) Randomized trial of an inhibitor of formation of advanced glycation end products in diabetic nephropathy. American Journal of Nephrology 24(1): 32–40. https://doi.org/10.1159/000075627 [PubMed]
Borghetti G, Von Lewinski D, Eaton DM, Sourij H, Houser SR, Wallner M (2018) Diabetic cardiomyopathy: current and future therapies. Beyond glycemic control. Frontiers in Physiology 9: 1514. https://doi.org/10.3389/fphys.2018.01514 [PubMed] [PMC]
Dedov II, Shestakova MV, Mayorov AY, Vikulova OK, Galstyan GR, Kuraeva TL, Peterkova VA, Smirnova OM, Starostina EG, Surkova EV, Sukhareva OY, Tokmakova AY, Shamkhalova MS, Jarek-Martynova IR, Beshlieva DD, Bondarenko ON, Volevodz NN, Grigoryan OR, Esayan RM, Ibragimova LI, Kalashnikov VY, Lipatov DV, Shestakova EA (2017) Standards of specialized diabetes care. Diabetes Mellitus [Saharnyj Diabet] 20(1S): 8–110. https://doi.org/10.14341/DM20171S8 [in Russian]
Dedov II, Shestakova MV, Vikulova OK, Zheleznyakova AV, Isakov MA, Sazonova DV, Mokrysheva NG (2023) Diabetes mellitus in the Russian Federation: dynamics of epidemiological indicators according to the federal register of diabetes mellitus for the period 2010–2022. Diabetes Mellitus [Saharnyj Diabet] 26(2): 104–123. https://doi.org/10.14341/DM13035 [in Russian]
Dedov II, Shestakova MV, Vikulova OK, Zheleznyakova AV, Isakov MA (2018) Diabetes mellitus in Russian Federation: prevalence, morbidity, mortality, parameters of glycaemic control and structure of glucose lowering therapy according to the federal diabetes register, status 2017. Diabetes Mellitus [Saharnyj Diabet] 2(3): 144–159. https://doi.org/10.14341/DM9686 [in Russian]
Ennis IL, Escudero EM, Console GM, Camihort G, Dumm CG, Seidler RW, Camilion de Hurtado MC, Cingolani HE (2003) Regression of isoproterenol-induced cardiac hypertrophy by Na+/H+ exchanger inhibition. Hypertension 41(6): 1324–1329. https://doi.org/10.1161/01.hyp.0000071180.12012.6e [PubMed]
Giacco F, Brownlee M (2010) Oxidative stress and diabetic complications. Circulation Research 107(9): 1058–1070. https://doi.org/10.1161/CIRCRESAHA.110.223545 [PubMed] [PMC]
International Diabetes Federation (2021) IDF Diabetes Atlas, 10th Brussels, Belgium. [cited 11 October 2025]. Available from: https: //www.diabetesatlas.org
Ivannikova EV, Smirnova OM (2019) The role of glycation end products and their receptors in the development of diabetes complications. Effective Pharmacotherapy. Endocrinology [Effektivnaya Farmakoterapiya. Endokrinologiya] 1(5): 20–26. [in Russian]
Jia G, Hill MA, Sowers JR (2018) Diabetic cardiomyopathy: an update of mechanisms contributing to this clinical entity. Circulation Research 122(4): 624–638. https://doi.org/10.1161/CIRCRESAHA.117.311586 [PubMed] [PMC]
Karayiannides S, Djupsjö C, Kuhl J, Hofman-Bang C, Norhammar A, Holzmann MJ, Lundman P (2021) Long-term prognosis in patients with acute myocardial infarction and newly detected glucose abnormalities: predictive value of oral glucose tolerance test and HbA1c. Cardiovascular Diabetology 20(1): 122. https://doi.org/10.1186/s12933-021-01315-5[PubMed] [PMC]
Kim J, Kim CS, Sohn E, Lee YM, Jo K, Shin SD, Kim JS (2014) Aminoguanidine protects against apoptosis of retinal ganglion cells in Zucker diabetic fatty rats. European Review for Medical and Pharmacological Sciences 18(11): 1573–1578. [PubMed]
Kochakian M, Manjula BN, Egan JJ (1996) Chronic dosing with aminoguanidine and novel advanced glycosylation end product-formation inhibitors ameliorates cross-linking of tail tendon collagen in STZ-induced diabetic rats. Diabetes 45: 1694–1700. https://doi.org/10.2337/diab.45.12.1694 [PubMed]
Li Q, Zhang J, Wang Y, Xu G (2009) Molecular spectral imaging system for quantitative immunohistochemical analysis of early diabetic retinopathy. Applied Spectroscopy 63(12): 1336–1342. https://doi.org/10.1366/000370209790108897 [PubMed]
Maltsev DV, Spasov AA, Kosolapov VA, Sultanova KT, Miroshnikov MV, Kotovskaya SK, Sapozhnikova IM, Rusinov VL (2020) Neurotoxicological properties of the compound AB-19 for prophylaxis and treatment of complications of diabetes mellitus. Toxicological Review [Toksikologicheskij Vestnik] 4: 39–43. https://doi.org/10.36946/0869-7922-2020-4-39-43 [in Russian]
Mironov AN, Bunatyan ND, Vasiliev AH (2012) Guidelines for Conducting Preclinical Studies of Medicinal Products. Part one. Grif and K Publishers, Moscow, 944 pp. [in Russian]
Miyauchi Y, Shikama H, Takasu T, Okamiya H, Umeda M, Hirasaki E, Ohhata I, Nakayama H, Nakagawa S (1996) Slowing of peripheral motor nerve conduction was ameliorated by aminoguanidine in streptozocin-induced diabetic rats. European Journal of Endocrinology 134(4): 467–473. https://doi.org/10.1530/eje.0.1340467 [PubMed]
Moldogazieva NT, Mokhosoev IM, Mel’nikova TI, Porozov YB, Terentiev AA (2019) Oxidative stress and advanced lipoxidation and glycation end products (ALEs and AGEs) in aging and age-related diseases. Oxidative Medicine and Cellular Longevity 2019(1): 3085756. https://doi.org/10.1155/2019/3085756 [PubMed] [PMC]
Nagai R, Murray DB, Metz TO, Baynes JW (2012) Chelation: a fundamental mechanism of action of AGE inhibitors, AGE breakers, and other inhibitors of diabetes complications. Diabetes 61(3): 549–559. https://doi.org/10.2337/db11-1120 [PubMed] [PMC]
Ovchinnikova MA, Markova TN, Shkodkina VV, Belyaeva OA (2025) Diabetic cardiomyopathy: definition, right to exist. Diabetes Mellitus [Saharnyj Diabet] 28(4): 387–396.https://doi.org/10.14341/DM13250 [in Russian]
Pokrovskii MV, Kochkarov VI, Pokrovskaja TG, Gladchenko MP, Artjushkova EB, Pashin EN, Brusnik MV, Chulukova TN, Kljavs JP, Korneev MM, Zelenkova TI, Malyhin VA, Belous AS, Zaloznyh JI, Majakov AI (2006) Methodical approaches for the quantitative estimation of development endothelial dysfunction at L-NAME-the induced model of deficiency of nitric oxide in experiment. Kuban Scientific Medical Bulletin [Kubanskij Nauchnyj Medicinskij Vestnik] 10: 72–77. [in Russian]
Qaed E, Aldahmash W, Mahyoub MA (2025) Advanced glycation end products (AGEs) and their role in diabetes mellitus and related complications: mechanisms and therapeutic insights. Glycoconjugate Journal 42(5-6): 209–223. https://doi.org/10.1007/s10719-025-10194-x[PubMed]
Rusinov VL, Chupakhin ON, Charushin VN, Sapozhnikova IM, Bliznik AM, Spasov AA, Petrov VI, Kuznetsova VA, Solovyova OA, Matsevich AI (2017) 4-Oxo-1,4-dihydropyrazolo[5,1-c]-1,2,4-triazine-3,8-dicarboxylic acid diethyl ester sodium salt, monohydrate: Patent RU 2612300 C1 Published 06.03.2017. [in Russian]
Singh VP, Bali A, Singh N, Laggi AS (2014) Advanced glycation end products and diabetic complications. The Korean Journal of Physiology & Pharmacology 18(1): 1–14. https://doi.org/10.4196/kjpp.2014.18.1.1 [PubMed] [PMC]
Spasov AA, Zhukovskaya ON, Rashchenko AI, Brigadirova AA, Litvinov RA, Gurova NA, Smirnov AV, Pan’shin NG, Abbas HSA, Morkovnik AS (2022) DF-5 compound delays development of diabetic nephropathy in rats. Pharmacy & Pharmacology 10(6): 549–561. https://doi.org/10.19163/2307-9266-2022-10-6-549-561
Tyurenkov IN, Perfilova VN (2012) Guidelines for preclinical studies of the cardiotonic activity of drugs. Guidelines for Conducting Preclinical Studies of Medicinal Products. Part one. Grif and K Publishers, Moscow, 375-384. [in Russian]
Wei M, Ong L, Smith MT, Ross FB, Schmid K, Hoey AJ, Burstow D, Brown L (2003) The streptozotocin-diabetic rat as a model of the chronic complications of human diabetes. Heart, Lung and Circulation 12(1): 44–50. https://doi.org/10.1046/j.1444-2892.2003.00160.x[PubMed]
Wong ND, Sattar N (2023) Cardiovascular risk in diabetes mellitus: epidemiology, assessment and prevention. Nature Reviews Cardiology 20(10): 685–695. https://doi.org/10.1038/s41569-023-00877-z [PubMed]
Yamagishi SI, Nakamura N, Suematsu M, Kaseda K, Matsui T (2015) Advanced glycation end products: a molecular target for vascular complications in diabetes. Molecular Medicine 21(S1): 32–40. https://doi.org/10.2119/molmed.2015.00067 [PubMed] [PMC]
Zhukovskaya ON, Spasov AA, Petrov VI, Raschenko AI, Litvinov RA, Morkovnik AS, Gurova NA, Smirnov AV, Schmidt MV, Pan’shin NG. Naumenko LV, Anisimova VA (2019) A drug with cardio-, nephro-, endothelio-, microangio-, macroangio- and encephaloprotective properties. Patent RU 2700791 C1 Published September 23, 2019. [in Russian]
Загрузки
Опубликован
Как цитировать
Выпуск
Раздел
Лицензия
Copyright (c) 2026 Gurova NA, Kosolapov VA, Smirnov AV, Babkov DA, Babkova VA, Taran AS, Panshin NG, Kotovskaya SK, Sapozhnikova IM, Rusinov VL, Spasov AA

Это произведение доступно по лицензии Creative Commons «Attribution» («Атрибуция») 4.0 Всемирная.
 Русский
Русский
 English
English

